
The penicillin-binding proteins (PBPs) synthesize and remodel the cell wall peptidoglycan, a major component of the bacterial cell wall that gives the cell its shape and rigidity –. This does not alter the authors’ adherence to PLOS ONE policies on sharing data and materials. All other funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.Ĭompeting interests: Jacques Dumas is employed by Sanofi-Aventis.


The specific role of this author is articulated in the ‘author contributions’ section. Sanofi-Aventis provided support in the form of salary for author JD, but did not have any additional role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript. Jacques Dumas is employed by Sanofi-Aventis. FK is research associates of the FRS-FNRS, Belgium. is recipient of a FRIA (Fonds de la Recherche pour l’Industrie et l’Agriculture) fellowship (F.R.S.-FNRS, Brussels, Belgium). P6/19), the Fonds de la Recherche Scientifique, (IISN 4.4505.09, IISN 4.4509.11, FRFC 2.4511.06F), the University of Liège (Fonds spéciaux, Crédit classique, C-06/19 and C-09/75), and a return grant to AD.
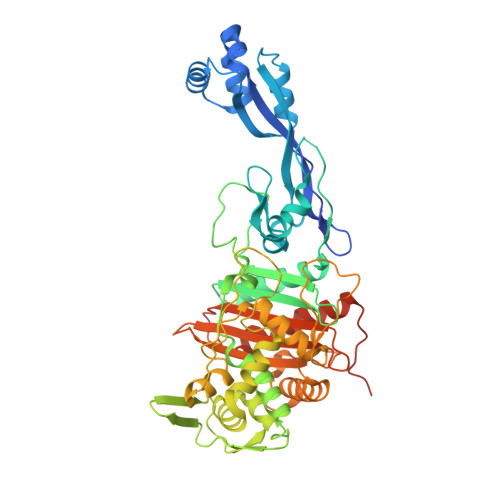
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.įunding: This work was supported in part by the Belgian Program on Interuniversity Poles of Attraction initiated by the Belgian State, Prime Minister’s Office, Science Policy programming (IAP no. Received: JanuAccepted: ApPublished: May 29, 2014Ĭopyright: © 2014 Sauvage et al. PLoS ONE 9(5):Įditor: Eric Cascales, Centre National de la Recherche Scientifique, Aix-Marseille Université, France (2014) Crystal Structure of Penicillin-Binding Protein 3 (PBP3) from Escherichia coli. These isolates were more frequent among non-invasive isolates.Citation: Sauvage E, Derouaux A, Fraipont C, Joris M, Herman R, Rocaboy M, et al. In particular, ftsI alleles that harboured the mutations D350N, S357N, M377I and S385T were resistant to ampicillin and third generation cephalosporins. BLNAR isolates were linked to modification in the ftsI gene encoding the penicillin binding protein 3 (PBP3). Moreover, the proportion of beta-lactamase negative ampicillin resistant isolates (BLNAR) was significantly higher among non-invasive isolates compared to that of invasive isolates (24% versus 7%, p < 0.001). Antibiotic susceptibility testing indicated that 24% of the invasive isolates were resistant to ampicillin but this percentage was significantly higher (51%, p < 0.001) among the non-invasive isolates. Non-typeable isolates showed extensive heterogeneity. WGS analysis suggested a serotype b to a capsule switching event. Serotype f was the most frequently observed but serotypes b and a were also present among invasive isolates. Most of the non-invasive isolates were non-typeable (99%) and this proportion was significantly less among invasive isolates 75%, p < 0.0001).

